Alison Chubb, PhD, Product Scientist, 23andMe

Booth
Come find us on the exhibition floor!
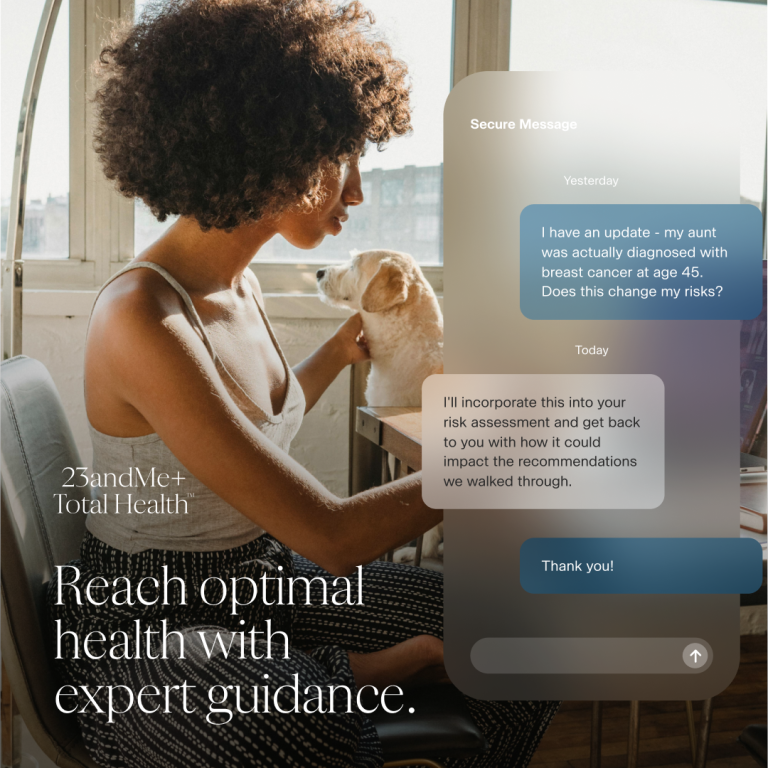
This is the third and final part of a blog series that delves into the different features included in Total Health and how they might apply to a healthcare professional. This blog focuses on clinical consults, how they work and the variety of benefits they provide for patients.
GeneReviews provides clinically relevant and medically actionable information for inherited conditions in a standardized, journal-style format, covering diagnosis, management and genetic counseling for patients and their families.
Genetics/Genomics Competency Center (G2C2) provides high-quality educational resources for group instruction or self-directed learning in genetics/genomics by healthcare educators and practitioners.
Find a Genetic Counselor assists physicians, patients and genetic counselors in accessing genetic counseling services.
CPIC© is an international consortium of clinicians and researchers that helps facilitate the use of pharmacogenetic information for patient care. CPIC evaluates the available clinical evidence supporting gene-drug pairs and publishes genotype-based dosing guidelines to help healthcare professional interpret genetic information for use in their prescribing practice.
PharmGKB is a pharmacogenetics knowledge resource that encompasses clinical information including guidelines and drug labels, potentially actionable gene-drug associations, and genotype-phenotype relationships. PharmGKB collects, curates, and disseminates knowledge about the impact of human genetic variation on drug responses.
DPWG is a collaboration between pharmacist, physicians, clinical pharmacologist, chemist, and others to develop therapeutic recommendation based on pharmacogenetic information and to help healthcare professionals integrate this information into their clinical practice.
Genetics Home Reference provides consumer-friendly information about the effects of genetic variation on human health.
23andMe Education modules explain key genetics concepts consumers need to know in order to get the most out of 23andMe reports.
Do You Speak BRCA? provides consumer-friendly BRCA education and information about 23andMe’s BRCA1/BRCA2 Selected Variants report, including helpful resources for educational and support services.
23andMe Help Center provides information about reports and tools as well as answers to any frequently asked questions. Customers can also contact our dedicated Customer Care Team here.
23andMe Blog posts relevant content in ancestry, health and traits, 23andMe research, 23andMe customer stories, news and announcements and more.
Questions? Contact us at medical@23andme.com.
*The 23andMe PGS test includes health predisposition and carrier status reports. Health predisposition reports include both reports that meet US FDA requirements for genetic health risks and the 23andMe Type 2 Diabetes health predisposition report which is based on 23andMe research and has not been reviewed by the FDA. The test uses qualitative genotyping to detect select clinically relevant variants in the genomic DNA of adults from saliva for the purpose of reporting and interpreting genetic health risks and reporting carrier status. It is not intended to diagnose any disease. Your ethnicity may affect the relevance of each report and how your genetic health risk results are interpreted. Each genetic health risk report describes if a person has variants associated with a higher risk of developing a disease, but does not describe a person’s overall risk of developing the disease. The test is not intended to tell you anything about your current state of health, or to be used to make medical decisions, including whether or not you should take a medication, how much of a medication you should take, or determine any treatment. Our carrier status reports can be used to determine carrier status, but cannot determine if you have two copies of any genetic variant. These carrier reports are not intended to tell you anything about your risk for developing a disease in the future, the health of your fetus, or your newborn child’s risk of developing a particular disease later in life. For certain conditions, we provide a single report that includes information on both carrier status and genetic health risk. Warnings & Limitations: The 23andMe PGS Genetic Health Risk Report for BRCA1/BRCA2 (Selected Variants) is indicated for reporting of the 185delAG and 5382insC variants in the BRCA1 gene and the 6174delT variant in the BRCA2 gene. The report describes if a woman is at increased risk of developing breast and ovarian cancer, and if a man is at increased risk of developing breast cancer or may be at increased risk of developing prostate cancer. The three variants included in this report are most common in people of Ashkenazi Jewish descent and do not represent the majority of BRCA1/BRCA2 variants in the general population. The MUTYH-Associated Polyposis Genetic Health Risk Report is indicated for reporting the Y179C and G396D variants in the MUTYH gene and an increased risk for colorectal cancer. The two variants included in this report are most common in people of Northern European descent. These reports do not include variants in other genes linked to hereditary cancers and the absence of variants included in these reports do not rule out the presence of other genetic variants that may impact cancer risk. The PGS test is not a substitute for visits to a healthcare professional for recommended screenings or appropriate follow-up. Results should be confirmed in a clinical setting before taking any medical action. For important information and limitations regarding other genetic health risk reports and carrier status reports, visit 23andme.com/test-info.
The 23andMe PGS test uses qualitative genotyping to detect 6 variants in 3 genes in the genomic DNA of adults from saliva for the purpose of reporting and interpreting information about the processing of certain therapeutics to inform discussions with a healthcare professional. It does not describe if a person will or will not respond to a particular therapeutic and does not describe the association between detected variants and any specific therapeutic. Results should be confirmed in a clinical setting with independent genetic testing before taking any medical action. Warning: Test information should not be used to start, stop, or change any course of treatment and does not test for all possible variants that may affect metabolism or protein function. The PGS test is not a substitute for visits to a healthcare professional. Making changes to your current regimen can lead to harmful side effects or reduced intended benefits of your medication, therefore consult with your healthcare professional before taking any medical action. For a complete list of the 6 variants tested, visit https://permalinks.23andme.com/pdf/pgt_product_info_page_21jan2020.pdf.